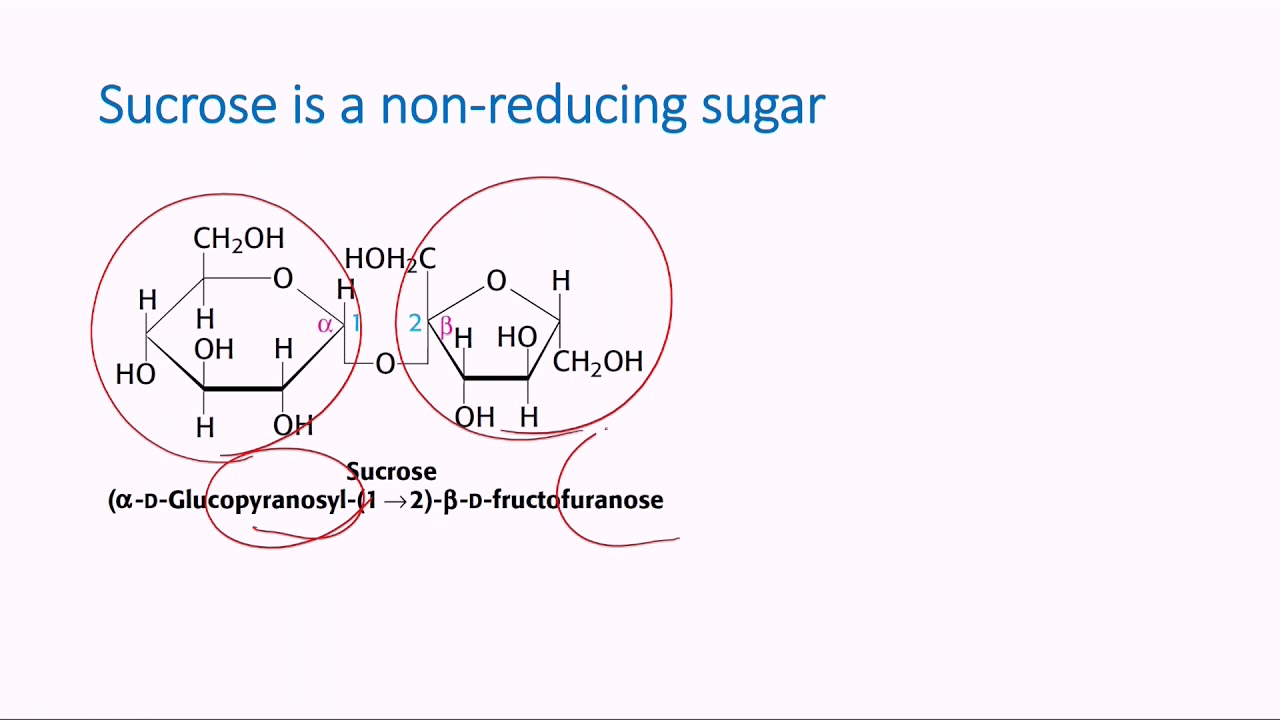
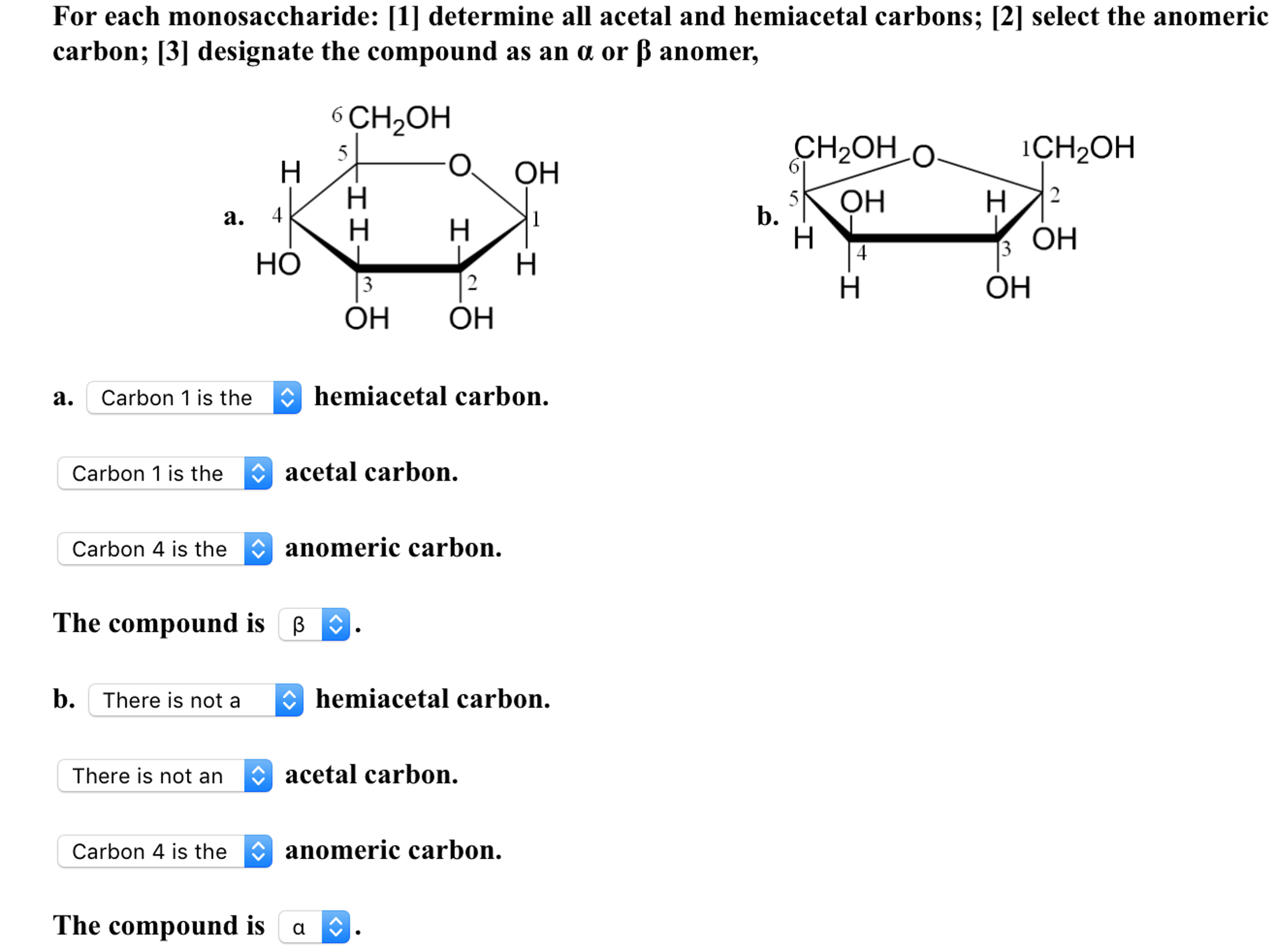
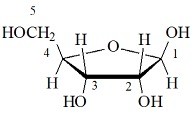
 Reducing sugars can be identified through the presence of a free anomeric carbon, meaning it is not in a glycosidic bond and has a free hydroxyl group. Sugars can also be described as being “non-reducing” or “reducing.” A reducing sugar is one that can act as a reducing agent. (The final ratios of each of these forms are usually affected by stereochemistry unique to each molecule.) This tendency to undergo spontaneous cyclization is known as mutarotation. As a result, many sugars can be found in various combinations of D-, L-, and linear forms. Note that as we convert the structure of the sugar from its two-dimensional to three-dimensional structure, the functional group that is pointing to the right of the Fischer projection will end up pointing downward in the Haworth projection.įor most sugars that are dissolved in a solution, this cyclization occurs spontaneously. A sugar is in its β-anomer form when the anomeric carbon’s substituent is on the same side of the plane as the highest numbered chiral center’s substituent. A sugar is in its α-anomer form when the anomeric carbon’s substituent is on the opposite side of the plane as the highest numbered chiral center’s substituent. In a cyclic sugar, the anomeric carbon is the carbon that has two bonds to oxygen.īe aware that a cyclic sugar can exist in two possible anomers: an ⍺-anomer and a β-anomer. While Fischer projections represent the straight-chain form of carbohydrates, you may also see sugars represented in their cyclic form as Haworth projections. If the hydroxyl group of the highest-numbered chiral carbon is on the left, it is in the L-configuration. If the hydroxyl group of the highest-numbered chiral carbon is on the right, the sugar is in the D-configuration. Image source: Chemistrylearner.For sugars, the absolute configuration is designated using D- and L- nomenclature instead of the R and S system used in organic chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
